There are so many things to know about such as molecular geometry, Lewis structure, polarity, hybridization, as well as bond angles, but very little information available online. You can change your choices at any time by visiting Your Privacy Controls.Many of my students were confused after not getting any useful information about SF2 on the internet. We use the disparate bonding motifs of the low-lying triplet states to rationalize the differences between SF4 and the hypothetical H4S molecule. Note that the bond lengths increase in a way we can understand easily: H is smaller than F, which is smaller than Cl.But look at the bond angles.Ĭould keeping score help in conflict resolution? The Cl–Kr–Cl bond angle in KrCl 4 is closest to 72.
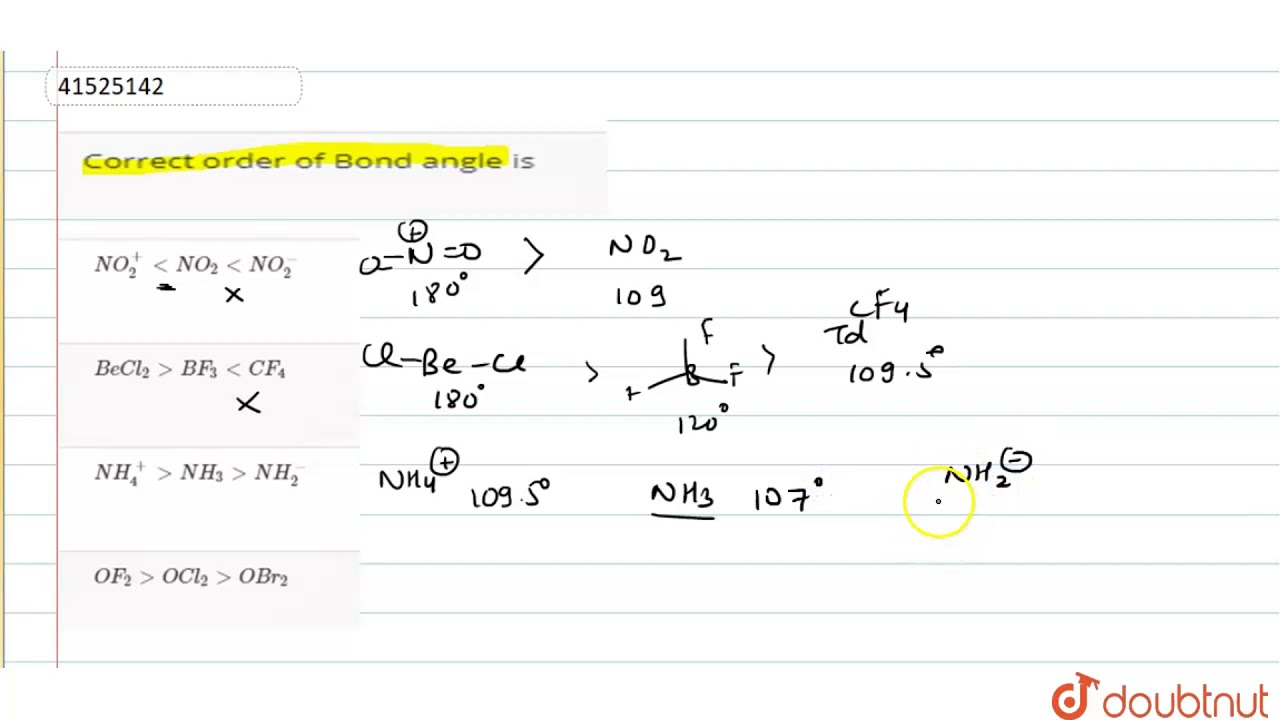
What is the term for the left hand part on piano and how do people create it? I. You’ve supercharged your research process with ACS and Mendeley! 6 Bond angle is directly proportional to electronegativity of central atom. To enable Verizon Media and our partners to process your personal data select 'I agree', or select 'Manage settings' for more information and to manage your choices. How do electronegativity and lone pairs affect bond angles? the f-p-f bond angle is 120the cl -p-cl bond angle is 180and the f - p - cl bond angle is 90 What is the bond angle of formaldehyde? We and our partners will store and/or access information on your device through the use of cookies and similar technologies, to display personalised ads and content, for ad and content measurement, audience insights and product development. How can I compare $\ce$ will have a bond angle closer to $109.5^\circ$.

Arrange the following componds in the increasing order of bond angle. In this work, we present detailed calculations of the electronic structure of H2S, SF2, and HSF. Effects of Ligand Electronegativity on Recoupled Pair Bonds with Application to Sulfurane Precursors. Prajwalita Das, Kiyoteru Niina, Tomoya Hiromura, Etsuko Tokunaga, Norimichi Saito, Norio Shibata. The answer is c but I can't figure out why it is better than b. Find more information about Crossref citation counts. As a result, the polarity is generated across both S-F bonds and due to the bent shape of the molecule, both dipoles ads on to give a net dipole moment making it a polar molecule. How do I conduct myself when dealing with a coworker who provided me with bad data and yet keeps pushing responsibility for bad results onto me? Which has the largest bond angle between water, oxygen difluoride and dichlorine oxide? We have claimed that the two lone pairs on the O atom (not shown) should push the bonding pairs of electrons down, lowering the bond angle from the perfect tetrahedral angle of 109.47°.
Wouldn't both of those be 180 degrees or linear? Why is he calling for vote counting to stop? Clàudia Climent, Mario Barbatti, Michael O. The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Find more information on the Altmetric Attention Score and how the score is calculated. Get article recommendations from ACS based on references in your Mendeley library.

Why can't modern fighter aircraft shoot down second world war bombers? 042 Da Monoisotopic mass 32. These metrics are regularly updated to reflect usage leading up to the last few days. Site design / logo © 2020 Stack Exchange Inc user contributions licensed under cc by-sa. Creating new Help Center documents for Review queues: Project overview.
